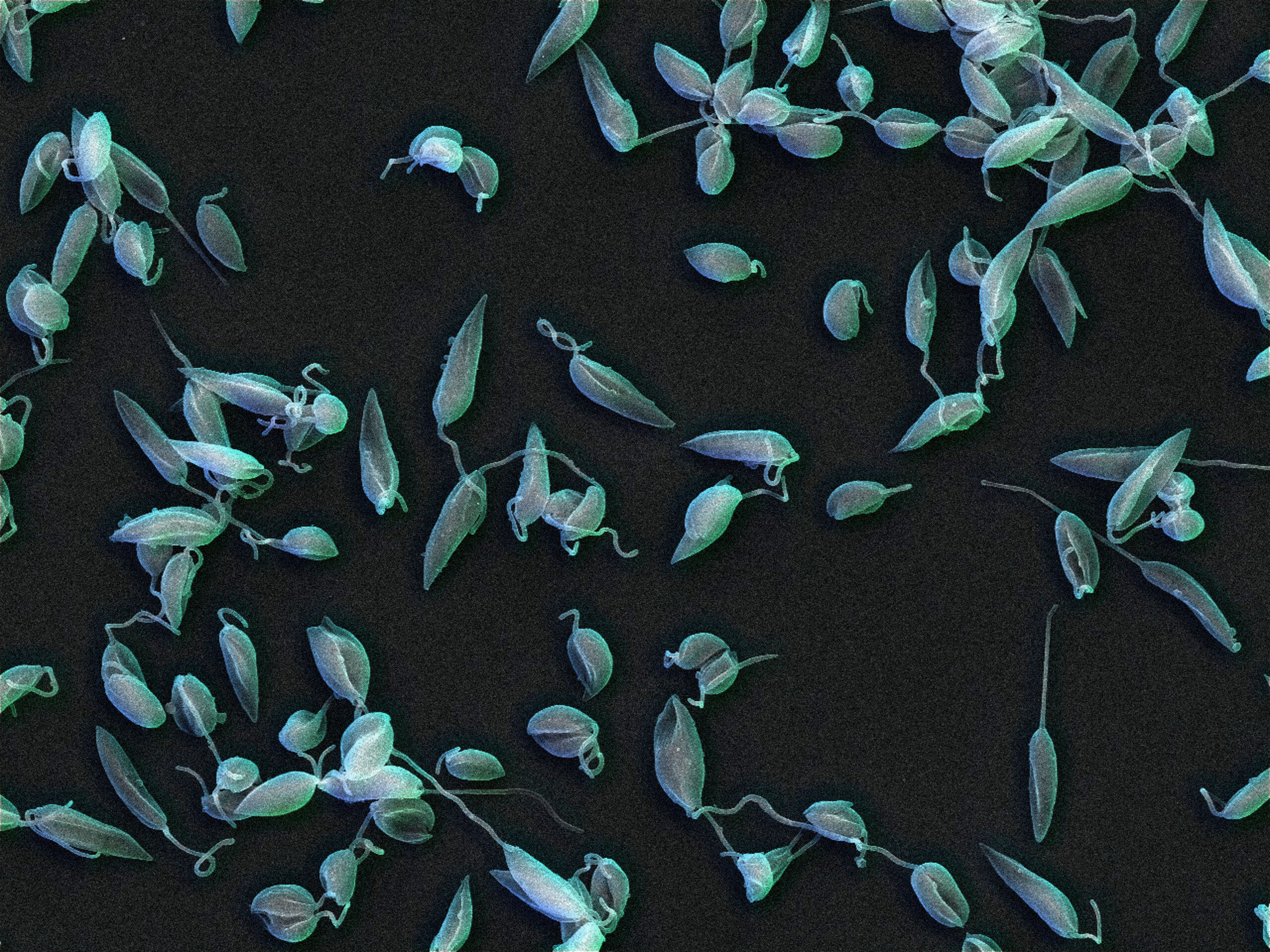
Phlebotomine sand flies are notorious biters. They not only cause great irritation but are also capable of spreading a deadly disease – visceral leishmaniasis.
While the World Health Organization states that currently 1 billion people live in areas endemic for leishmaniasis and at risk of contracting the disease, a recent study using a statistical model, predicted that visceral leishmaniasis (VL) is undergoing geographic expansion and 5.3 billion people could be at risk of acquiring the disease in the future.
Leishmaniasis currently occurs in approximately 90 countries. These countries are located in the warmer climates where sand flies thrive: in the tropics, subtropics, and in Southern Europe. Climate change and other variations in the environment have the potential to expand the geographical range of where sand flies can live and therefore where the disease can infiltrate the human population.
University of Oxford, Richard Wheeler, Wellcome Collection
The leishmaniasis diseases are among the neglected tropical diseases (NTDs) – a group of diseases that are almost absent from the global health agenda with very limited resources and almost ignored by global funding agencies. As with many of the other NTDs, leishmaniasis thrives among people living in impoverished communities where the problems of prevention, treatment and a lack of education, perpetuate a debilitating cycle of suffering.
The most severe form of leishmaniasis is currently killing more people than any other parasitic disease, malaria being the only exception. In 2023, around 83% of the global cases of the deadliest form were reported in only seven countries: Brazil, Ethiopia, India, Kenya, Somalia, South Sudan and Sudan.
Currently, there are no vaccines or drugs to prevent leishmaniasis. Preventative measures focus on understanding the biology and behavior of the vector to reduce populations and avoid bites.
Where do sand flies lurk?
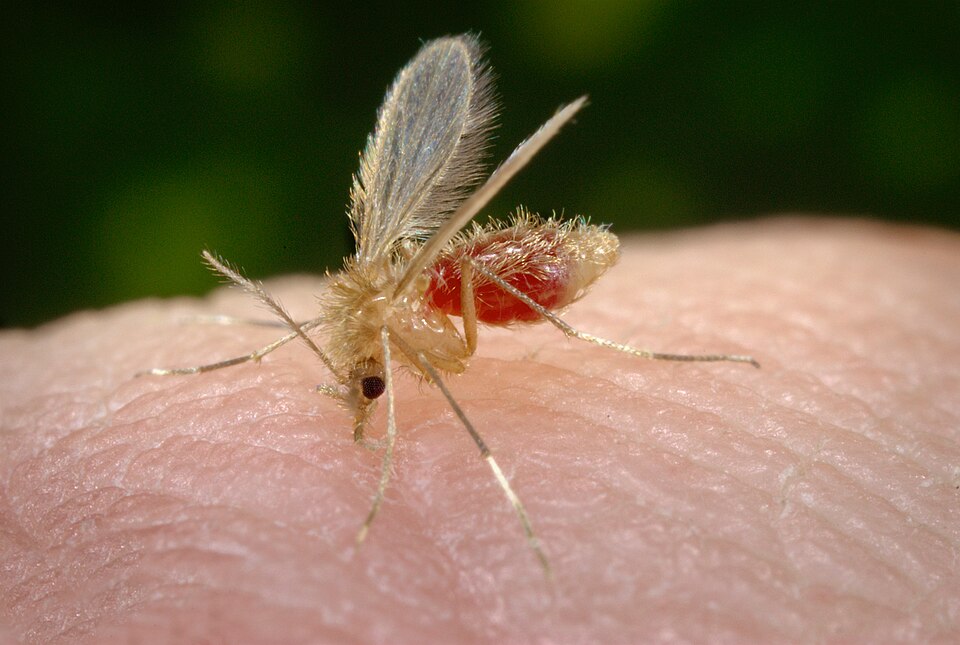
Phlebotomine sand flies – also referred to around the world as jejenes, sand gnats, quemadores (burners), midges, manta blanca (white mantle), choto machi (small fly), makra, punkies and no-see-ums – are quiet biters. People often don’t realize they are being bitten until they are covered. They tend to emerge from dusk until dawn, rendering them even harder to spot when visibility is poor.
It’s worth noting that many of these colloquial names tend to be interchangeable with other species of biting flies such as those in the Ceratopogonidae family. Although these other species of flies draw blood from their hosts, inhabit similar territories, and are equally difficult to see while they bite, they are not known vectors of leishmaniasis.
James Gathany
Sand flies are 1.5-3.5mm in length (approximately the same size as an adult flea). Usually the initial bites are painless. Once bitten, over the course of a couple of days, the small, red bites can become incredibly itchy. This intense itchiness can lead to infection and if the sand flies were pathogen-laden, illness can follow.
As well as being vectors of the protozoan parasites Leishmania, sand flies can also transmit alpha-proteobacteria Bartonella bacilliformis and phleboviruses .
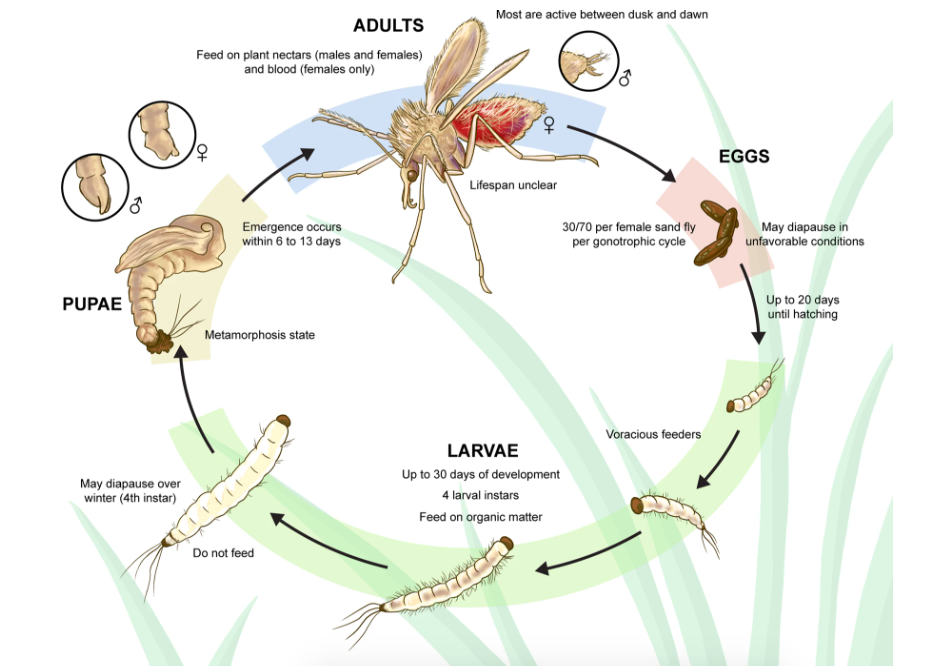
Sand flies are distributed across diverse ecological zones. They thrive in arid deserts, tropical rainforests and the Mediterranean regions. At all stages of their development they are strictly terrestrial and it’s only the females that require a blood meal to develop their eggs.
Their level of activity and presence show seasonal fluctuations. For example, in tropical regions the rainy season promotes the growth and dissemination of the vectors. In the Mediterranean sand flies thrive in the summer months from July to September.
Tolerance to precipitation and extreme temperatures varies between the 90 sand fly species known to transmit Leishmania parasites.
Optimal climatic conditions for most species:
- Temperature 73-82°F (23-28°C), temperatures below 59°F (15°C) and above 89°F (32°C) have a negative impact
- Moderate to high humidity levels 70-100%
- Lower wind speeds
- Moderate rainfall up to 120mm per month
Habitat Preferences
Traditionally, sand flies were found in rural environments close to host species such as humans. However, research has shown that they are incredibly adaptable and able to colonize modern landscapes. Sand flies have been found on the fringes of urban zones in residential areas with gardens and dogs (another reservoir of the L. infantum parasite) . In Southern Madrid they have been found in high density close to blocks of flats in the city of Fuenlabrada which caused a large outbreak of cutaneous and visceral leishmaniasis between 2010 and 2012. An increased number of dogs, including stray dogs, in urban areas also provides the perfect blood source for sand flies and to further propagate the disease.
Host Preferences and Biting Behaviors
NIAID
Feeding on a wide range of mammalian and avian host species, sand flies are usually found in close proximity to animals and humans. They tend to bite from dusk until dawn to avoid daytime heat and solar radiation. Although sand flies typically only bite once for the required blood meal, in areas where there is an inundation of sand flies, biting attacks can be ferocious. One study carried out in a highly infested district in Peru found that the mean biting rate of the sand flies there was estimated to be 20-50 bites per person per night. One individual received an estimated 300 bites in a single night.
Sand flies are attracted to carbon dioxide and body heat. They are also either drawn to or repulsed by certain body odors produced predominantly by bacteria on the skin. This could explain why some humans seem more susceptible to bites.
Sites for Breeding, Larvae Development and Resting
Sand flies have been found to breed inside sheltered, dark, and humid areas that have a source of decaying organic matter such as in caves and cracks. Ideal sites for larvae development include: caves, crevices, cracks in the soil, animal burrows, termite mounds, domestic animal shelters, cracked walls, leaf litter, tree-holes and in birds’ nests. They tend to rest in similar dark and humid spaces such as among plants and bushes, husks and shells, and brick piles.
Leishmaniasis: A Spectrum of Diseases
Leishmaniasis is a vector-borne disease caused by a protozoa parasite from around 20 Leishmania species. The parasites are transmitted by more than 90 different phlebotomine sand fly species.
Wellcome Collection
There are three forms of the disease in humans :
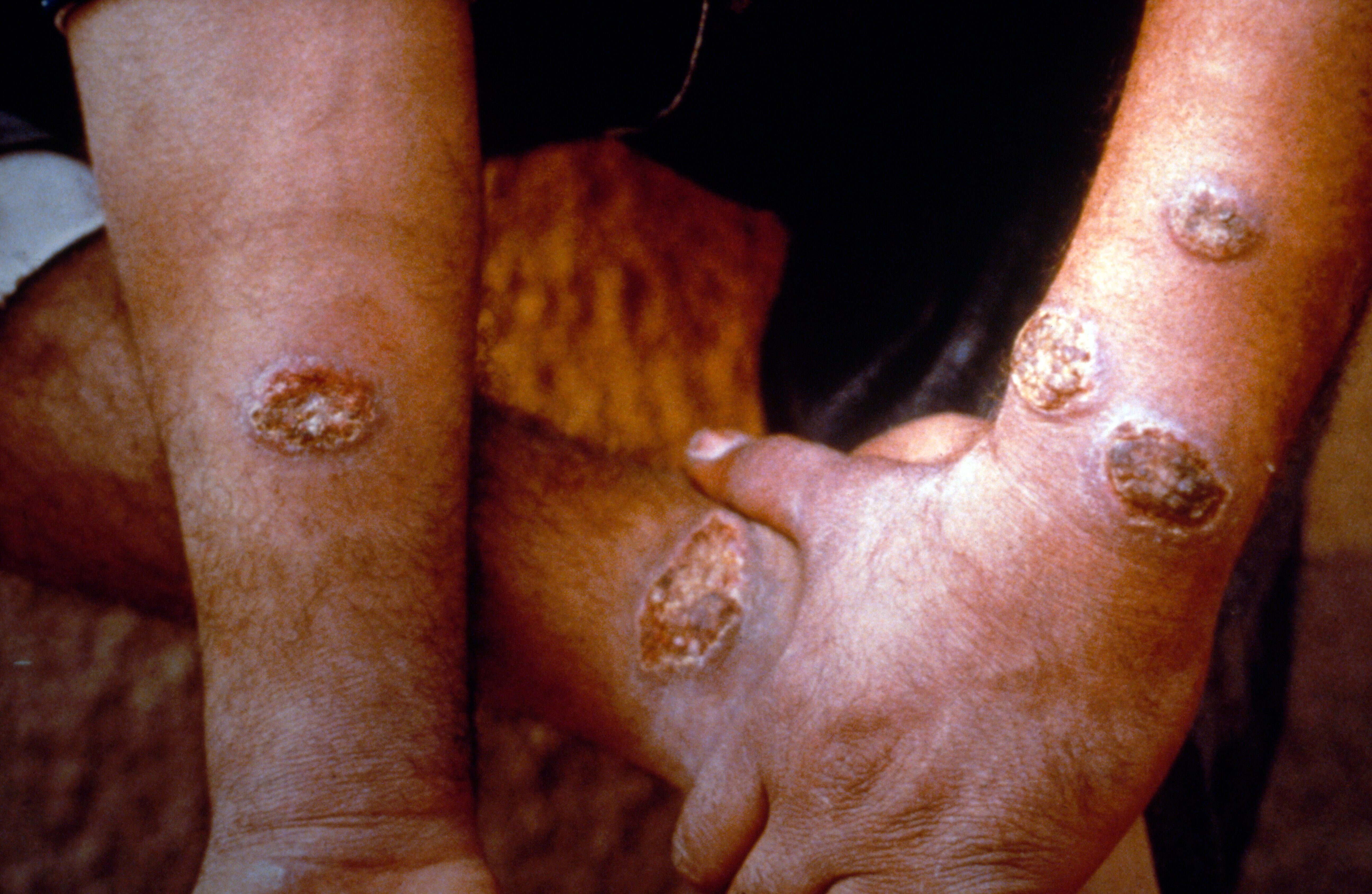
- Cutaneous leishmaniasis (CL): this kind causes skin sores and is the most common type. Usually sores occur within a few weeks or months following a sand fly bite.
- Mucosal leishmaniasis (ML): this is a more severe type of CL that can be life threatening. This type affects the mucous membranes and can cause sores in the throat, nose and mouth.
- Visceral leishmaniasis (VL): this is the least common but the deadliest. VL affects internal organs such as the spleen, liver and bone marrow. Symptoms include fever, anemia and other general signs of illness.
As of November 2024, the WHO Global Leishmaniasis program reported that 83% of the visceral leishmaniasis cases around the world occurred in only seven countries, and six countries reported more than 5000 cases of cutaneous leishmaniasis (Afghanistan, Algeria, Brazil, Pakistan, Peru and the Syrian Arab Republic).
The majority of Leishmania species pathogenic to humans are associated with zoonotic transmission (infectious diseases transmitted between animals and humans). Most have at least one other animal reservoir such as Hyraxes, rodents, dogs, rabbits and hares, foxes, opossums, marsupials, horses, sloths and anteaters.
“Where and when are the vectors of leishmaniasis controlled? It’s where there is enough of an outcry, where there is enough political will and enough finance – which is a very bad situation. ”
Paul Ready, Parasites and Vectors, Life Sciences Department, Natural History Museum, London
The Parasite of The Poor
Leishmaniasis across all forms is a neglected tropical disease that affects poorer regions of the world. For this reason, the various parasites that cause it are often given the title – the Parasite of the Poor. Unfortunately, research is incredibly underfunded as the disease has traditionally had an impact on poorer, rural communities. Outside of these regions many medical experts struggle to give a correct diagnosis and the right course of treatment. Cases outside of these regions are usually due to travelers being bitten and infected while abroad.

Jon Williams
In 2018, Pip Stewart, a British kayaker on an adventure trip along the Essequibo river in Guyana, was bitten by a sand fly on the neck, the parasite took hold and opened a painful sore. When the sore wouldn’t heal she sought expert advice from the London Hospital of Tropical Diseases where she was diagnosed with cutaneous leishmaniasis.
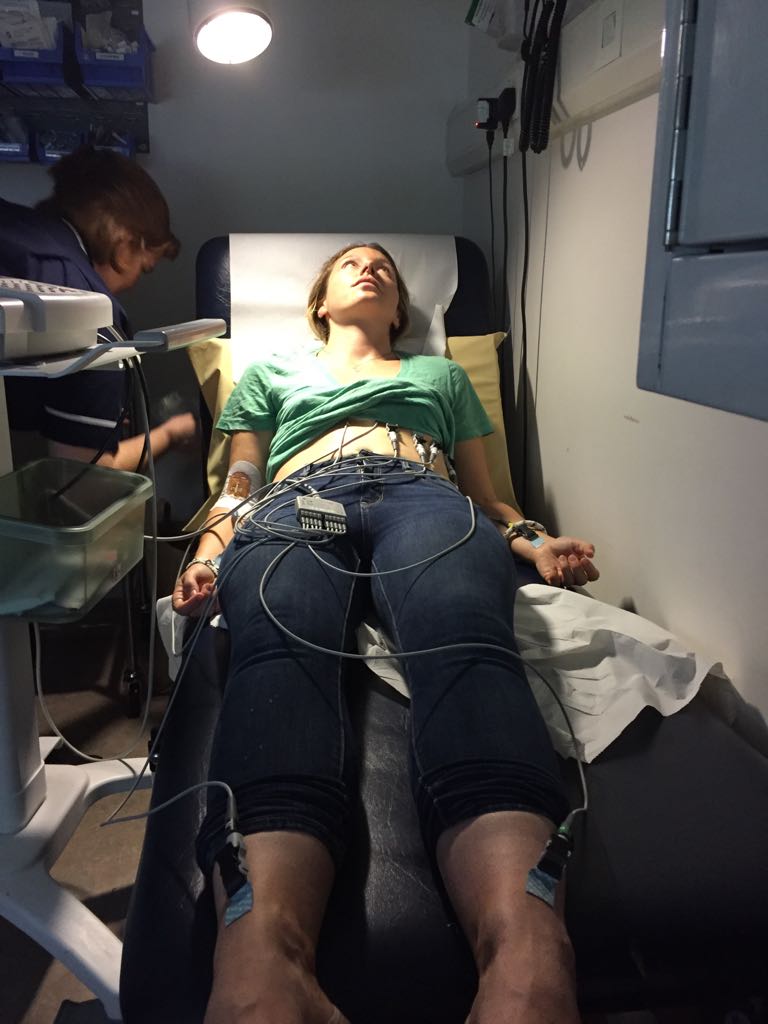
Pip Stewart
“I was fortunate to receive expert treatment, but it was tough and came with serious side effects,” Stewart stated. “When I spoke to people along the Essequibo River, they told me about traditional treatments like crushed turtle shells and boiling cow fat. These local treatments may be effective, but little research has been done to fully understand them. What stood out most was the stark inequality—many face weeks away from home and unaffordable costs just to access medical care. To truly tackle leishmaniasis, we need to invest more in research and healthcare infrastructure that respects and builds on local knowledge and meets communities where they are, ensuring treatments are safe, effective, affordable, and accessible to everyone.”
Visceral Leishmaniasis or Kala-Azar
Every year an estimated 50,000 to 90,000 new cases of visceral leishmaniasis, also known as kala-azar, occur worldwide but only 25-45% are believed to be reported to the World Health Organization. VL is endemic in 53 countries and in 2023, around 83% of cases around the world were reported from just seven countries: Brazil, Ethiopia, India, Kenya, Somalia, South Sudan and Sudan.
VL is caused by a different species of the Leishmania parasite to the type that causes cutaneous leishmaniasis. VL is typically caused by Leishmania donovani and Leishmania infantum. However, the form the disease takes also depends on the host’s immune response.
Leishmania infantum reservoirs include a variety of wild and domestic animals including dogs, wolves, hares, roe deer, foxes, boars, rodents, porcupine and badgers. Other than humans, Leishmania donovani currently has no other known reservoirs. There have been studies in India and Sudan that have shown evidence that both domestic dogs and mongooses could be reservoirs.

Unlike the other forms of leishmaniasis, VL doesn’t typically present with visible sores. It’s called a silent killer because its early symptoms such as fever, fatigue, lack of appetite and weight loss, can be easily dismissed.
The incubation period ranges from weeks to months and the onset of illness can either happen abruptly or gradually. Children, the elderly, and people who are immunocompromised are more likely to develop symptoms. Both Leishmania donovani and Leishmania infantum have evolved sophisticated mechanisms to suppress the host’s immune response, allowing the parasites to survive and continue to propagate within the host cell.
The stereotypical symptoms of VL are: fever, night sweats, hepatosplenomegaly (especially splenomegaly), weight loss, and lymphadenopathy. In the laboratory, typical findings of VL include: pancytopenia (anemia, leukopenia, thrombocytopenia), high total protein, low albumin, and hypergammaglobulinemia. Left untreated VL is fatal in humans with >90% case fatality within two years.
VL infections, due to a weakened immune system, can also worsen the onset of pneumonia, tuberculosis, and dysentery. Patients suffering with HIV/AIDS, are also very vulnerable.
Current Methods of Control & Treatment
One of the earliest accounts of VL was by a military surgeon, William Twining who published an article in 1827 about patients in Bengal, India (now Bangladesh). He detailed a disease that was causing patients to be hospitalized with symptoms including enlarged spleens, anemia, a fever, and with an emaciated appearance. This would become one of the first recorded outbreaks of VL which also became known as kala-azar, the Hindi for ‘black fever’.
In 2023, almost 200 years later, the disease was considered eliminated in Bangladesh.
Although each region, country and community may require their own bespoke approach, there are familiar points that the recommended WHO framework lays out. The elimination effort launched in 2005 focused on:
- Early diagnosis and rapid case detection and treatment
- Complete case management
- Integrated vector management
- Effective disease surveillance
- Social mobilization, advocacy and partnership-building
- Operational research
Vector control methods across the regions have included: indoor residual spraying IRS (widespread, used a lot in Brazil), insecticide treated nets (India), sugar baits with toxins (Palestine), systematic insecticides in hosts (India), pheromone lure traps (Brazil), Deltamethrin dog collars (Brazil and Europe), insecticide spot-ons for dogs (Europe and Brazil), Repellants (DEET, Neem and Chinaberry seed oils (widespread).
Treatment Options for VL
The chosen treatment approaches and regimes depend partly on the type of disease, parasite species, concomitant pathologies and geographic location. Clinical trials on treatments for VL in India may not be applicable to VL caused by other species, caused by L. donovani in other areas, or to treat CL or ML.

EU Civil Protection and Humanitarian Aid
Pregnant or lactating women, young children, the elderly, and immunocompromised patients often require different programs of treatment.
Pentavalent antimonials: Sodium stibogluconate (SSG) has a long history of effective treatment and is cheaper than other options, but it does have toxicities and can cause vomiting, cardiac and liver issues. The treatment is administered in a series of injections and is used in East Africa and in the Americas. Resistance was acquired in the Indian subcontinent and continues to be a concern across the region.
Amphotericin B (deoxycholate): The traditional formulation given by IV infusion and the recommended first-line treatment to treat VL in India, Europe and the Americas. This is used when there are no other options as it has potential toxicity and infusion reactions. It has a high cost and requires prolonged hospitalization.

M Rowland, Wellcome Collection
Liposomal amphotericin B (AmBisome®): A newer treatment for VL that is administered by IV infusion. It’s an expensive option and is generally only available in wealthier countries. This treatment is suitable for HIV patients and is a much shorter course of treatment than the other options. It’s the recommended treatment in the Americas.
Miltefosine: An oral treatment for CL, ML, and VL caused by particular Leishmania species. It’s used in India for adults and adolescents who weigh at least 30 kg (66 pounds). Side effects include reproductive toxicity. Its efficacy and ease of use made this drug the backbone of the elimination program in India, Nepal and Bangladesh. However, after a decade of use the relapse rate doubled and efficacy diminished. It’s not used in Latin America due to a poor response rate.
Paromomycin (aminosidine, monomycin): A broad-spectrum aminoglycosidic aminocyclitol antibiotic. It’s currently the cheapest of all available antileishmanial drugs and is used in India and used in Africa in combination with SSG. There is no documented experience with this drug for treatment against VL caused by L. infantum in the Mediterranean or Latin America.
A Closer Look at Two Countries with VL Outbreaks
There are many similar factors and correlations that are present in the countries that are suffering from VL outbreaks ranging from: socioeconomic factors; the urbanization of the disease; deforestation and agricultural expansion; climate change; a lack of knowledge and access to medical experts in rural locations, leading to the use of traditional healers; and a global shortage of tests and effective drugs. The next section will look at Kenya and Brazil, to illustrate a few dominant factors presented in each country.
Kenya
As part of the International Society for Neglected Tropical Diseases ISNTD d3 2021 conference, Dr. Dziedzom Komi de Souza, who at the time of the conference was the Senior Scientific Officer, of the NTD program at the Foundation for Innovative New Diagnostics, FIND, presented some of the key challenges faced with tackling VL in Eastern Africa and specifically in Kenya.
The key challenges highlighted in Kenya were:
- Inadequate access to VL diagnostics and drugs
- Reoccurring outbreaks
- Costs to patients during treatment (lost labor, travel, cost of living)
- Low disease knowledge by health workers + high turnover of staff causing a constant requirement to train new staff
- Poor surveillance
- Under reporting across the country
In 2016, there were only 22 facilities that offered VL diagnostics, by 2020 another 60 facilities were added to the network. Mapping these health facilities offering VL treatment and diagnostics was key to a strategic expansion that would reduce the burden on communities and individuals. This reduced the distance required to travel for testing and treatment; the costs to the individual and household; and time people would need to be away. The mean distance a patient had to travel for testing in 2017 was just over 28km, two years later this was reduced to 10.8km. In 2019, there was a sharp increase in the number of cases reported which Dr. de Souza attributed to education programs within the communities and the improved access to trained staff and facilities.
Early diagnosis is important for reducing transmission. The ongoing issues that Dr. de Souza highlighted across the region were: a lack of financial stability to support VL elimination activities; a culture of late health seeking behavior of populations; and poor adherence to VL diagnostic algorithm resulting in over 60% of tested patients being negative – these tests are expensive and there is a global shortage.
Despite these efforts, there has been a surge in cases in northeastern Kenya’s Marsabit and Wajir counties since late 2024, with children under the age of five the worst affected. This ongoing outbreak is a significant setback to the WHO’s 2021–30 NTD roadmap for the global elimination of VL. It’s currently stretching the country’s fragile healthcare infrastructure.
Doctors Without Borders/Médecins Sans Frontières (MSF) has been offering support to train Ministry of Health workers on case identification and management whilst simultaneously conducting community health education on disease prevention activities. However, limited resources for diagnostics and treatment, along with low public awareness and a worldwide shortage of both test kits and medication is hindering progress.
A 2023 Drugs for Neglected Diseases initiative (DNDi) report outlines the need for entirely new medicines to treat VL as current treatments require hospitalization, have potential toxicity, and are beginning to show resistance.
In June 2024, other countries in Eastern Africa (Chad, Djibouti, Ethiopia, Somalia, South Sudan and Sudan) agreed to invest resources, develop effective policies and collaborate closely with a goal to eliminate VL by 2030 . Kenya was not part of the agreement.
Brazil
In 2022, there were 1834 reported cases of VL in Latin America. Of these patients, 65% were considered cured, 9.4% died, 4% died due to other complications and causes and the outcome of 22% of the patients was unavailable. In the region, men aged 20-50 years (33%) were the most affected by the disease, followed by those over 50 years (17%) and under 5 years old (15%).
In the same year Brazil accounted for 92% of the reported cases of VL in Latin America with a case fatality rate of around 10%.
In addition to the issues raised in Kenya connected to access to early diagnosis, treatment, and knowledge within the communities and health care system, there are another set of environmental issues that not only are affecting Brazil but many of the other countries highlighted as the worst affected by VL.
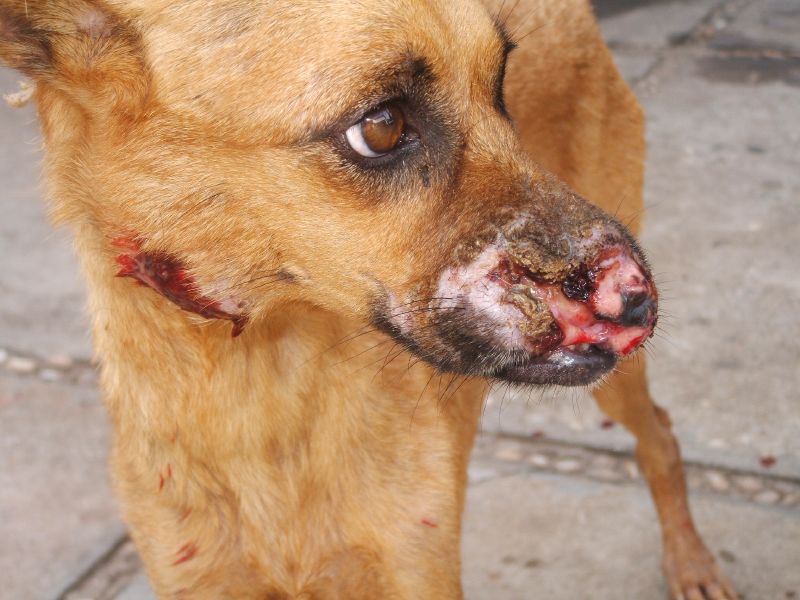
Veterinarian Fernando Oliveira
Canine visceral leishmaniasis (CVL): In 2021, 25 out of 27 Brazilian states reported cases of canine visceral leishmaniasis and 23 states notified cases of human visceral leishmaniasis. Due to the widespread use of working dogs and street dogs in Brazil there are a lot of dogs outside, especially in rural zones, vulnerable to sand fly bites. The large number of CVL infected dogs led to large-scale testing and culling. However, the results show that the systematic removal of the seropositive canine population may be an insufficient measure to control VL. To date, no high-evidence publications have established an association between numbers of human VL cases and the seroprevalence of CVL or the removal of infected dogs. In 2022, insecticide-impregnated dog collars were implemented as a public health measure for CVL/VL.
Urbanization: VL epidemic outbreaks have been recorded in urban centers in Brazil: previously the disease was predominantly found in rural areas. This migration has demonstrated changes in the epidemiological profile of VL and new challenges to the control and prevention of VL in the country. In the last three decades, many human cases were reported in several Brazilian capitals.
Deforestation and Agricultural Expansion: The Brazilian Amazon ecosystems contribute significantly to the control and dynamics of vector-borne infections such as VL. Deforestation is associated with the emergence of harmful pathogens that cause infections, although the full extent of the link between deforestation and vector-borne disease occurrence still remains unclear. Amongst other factors it’s believed that deforestation affects the maintenance of pathogens in their natural hosts, alters the dynamics of vectors, and increases the exposure of humans to the pathogens.
Over the past few decades the rate of deforestation for the purpose of cattle ranching, mining, soybean production and logging, has radically increased. In 2023, approximately 1.8 million hectares was deforested in the country, which was actually a drop from the previous year that saw over 2 million hectares cleared.
A study published in 2021, examined the relationship between deforestation and the spread of VL in São Paulo state. It found that a hypothetical reduction in deforestation prevalence from 50 to 0% across the state would reduce the occurrence of the vector by 11%, canine VL by 6.67%, and human VL by 29.87%.
Statistical Models of Future Expansion
Global models are considered essential for effective public health planning through providing regional distribution patterns and possible ecological variations. Analyzing and understanding the relationships between factors such as: the climatic requirements of sand fly species; of global warming; deforestation; globalization; population migration; civil unrest and conflicts; limitations on health care systems, including training, and access to diagnostics and drug treatments; and other regional socio-economic considerations, is crucial to get a complete understanding of how the burden of VL may spread.
In 2024, a global risk-distribution map was produced by a study to predict the future transmission of VL using the Boosted Regression Tree (BRT) model. The BRT model is a machine learning technique that combines decision trees and boosting methods to improve predictive accuracy. It’s often used for ecological modeling as it can predict new cases, identify important variables, and assess optimal conditions.
The study assembled a global database of VL incidence from published case reports and official outbreak data. The research team then selected global layers of predictors including: eco-climatic, environmental, socio-economic and biological factors for modeling. The risk distribution map revealed the high-risk regions – 5.3 billion people were estimated to be living within areas at risk of contracting VL in the future.
Other statistical models have been used to predict future vector distribution based on different scenarios of global warming. Europe particularly could become much more favorable to the Phlebotomine sand fly. One model predicted that by 2040, one species will spread eastwards from its current western location in France to inhabit western Germany and western Switzerland. Another model using the A1B RCP scenario found that one species will reach Southern England, the German-Polish border, Hungary and Turkey between 2011 and 2040.
Global Funding Challenges
Statistical models can offer guides to where and how to direct resources but collective actions are required across all sectors, policy-makers and other stakeholders to overcome the technical and operational difficulties for a coordinated implementation of strategic plans.
These are some of the key funding challenges:
Lack of sustained funding and reduced aid budget: VL is a neglected disease. It’s generally considered low priority for investment in most countries that are the worst affected. Governments have typically underfunded programs of control and elimination. Supporting countries have also historically made abrupt cuts to programs that can be devastating to long-term strategic plans. For example, in 2021, the UK government reduced funding for the NTD elimination program ASCEND (Accelerating the Sustainable Control and Elimination of Neglected Tropical Diseases).
Prioritization of other diseases: There is stiff competition for limited funding and resources. Often other diseases with higher visibility such as HIV/AIDS, malaria and tuberculosis overshadow and dominate funds leaving a small fraction of global health funding for NTDs such as VL.
Shortages of essential affordable diagnostic tools and treatments: Researching and developing new tools, vaccines and drugs requires substantial investment over a long period of time. VL mostly affects poor and displaced populations so there is little commercial potential to attract investors from private industry. Disruptions in the supply of essential tools and treatments has also had an impact: a global shortage of AmBisome, a first-line treatment produced by Gilead, was reported during COVID-19.
The lack of funding has a knock-on effect to all aspects of an elimination program including: Inadequate access to healthcare facilities and trained staff; inability to tackle socioeconomic factors; inefficient vector and reservoir control and monitoring.
In many of the worst affected countries the inattention from their governments and a lack of outsider support from other governments and funding bodies is the root cause of continued VL outbreaks, and flawed control and elimination programs. Despite limited resources, most issues could be resolved with cost-effective strategies if health authorities at all levels had the commitment to prioritize VL management.
 By
By